Lab Research Overview Video
Atmospheric Chemistry Career Video
Atmospheric chamber studies
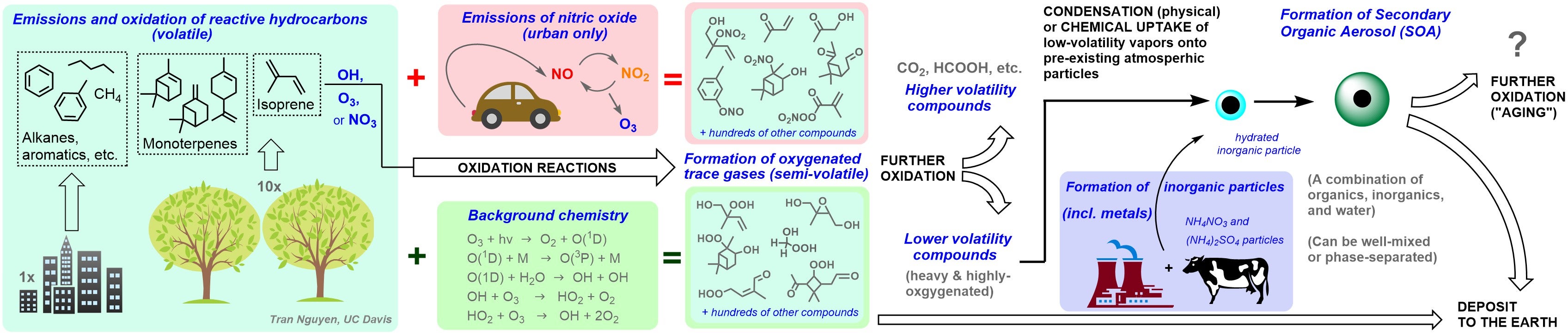
Oxidation is the main fate for the hydrocarbons that are emitted to the atmosphere. Depending on whether the oxidation chemistry occurs in a clean or urban-influenced environment, the reaction products can vary from carbon dioxide to tropospheric pollutants like ozone and secondary organic aerosols. In addition to their effects on air quality (leading to poor visibility and adverse human health), aerosols are a large uncertainty in the understanding anthropogenic climate change.
We use a 10 cubic meter Teflon reaction chamber to study oxidation chemistry in a simulated atmosphere. The chamber is equipped with 72 UV black lamps to perform photochemical reactions and a capability to control temperatures from -20 to 60 Celsius. We introduce hydrocarbons into the chamber and generate oxidant species (ozone, OH radicals, NO3 radicals) to initiate the reaction. Reactions can be performed in the presence and absence of NOx, seed particles, or relative humidity (<5% to 90%).

Reactions in the chamber are monitored by a suite of in-situ instrumentation: (a) temperature and relative humidity probe, (b) GC with flame ionization detector, (c) NOx chemiluminescence analyzer, (d) ozone photometric analyzer, (e) chemical-ionization mass spectrometer (below), and (f) scanning mobility particle sizer.
In addition, our lab has the following capabilities to analyze samples that have been collected, e.g., aerosols on a filter or a gaseous mixture in a canister: (a) GC with mass selective detector, (b) high-resolution mass spectrometry (below), (c) LC-MS, (d) HPLC-UV, (d) UV-Vis spectroscopy, and (e) FT-IR spectroscopy with 10-m cell.
Chemical ionization mass spectrometry (CIMS)
We use a custom-built chemical ionization mass spectrometer with triple-quadrupole mass analyzer that is modeled after the CIMS developed by the Wennberg group at Caltech. The CIMS operates using the CF3O- reagent ion and can detect a large range of volatile organic compounds from the following classes: (1) H2O2 and organic hydroperoxides, (2) acidic compounds such as SO2, HCN, HNO3, and organic acids, (3) polyols (glycol, methyltetrols), (4) hydroxy epoxides (e.g., IEPOX), (5) hydroxy carbonyls (e.g., glycolaldehyde, hydroxyacetone), (6) hydroxy nitrates (e.g., isoprene and pinene nitrates), and (7) other multifunctional compounds (e.g., nitrooxy hydroperoxides). We are in the process of building a CIMS using an Ionicon time-of-flight mass analyzer.

High resolution mass spectrometry (HRMS)
We use a custom-built nano-electrospray ionization (ESI) source that is switchable to a nano-desorption electrospray (nano-DESI) source, modeled after the nano-DESI design that was developed in the lab of Alexander Laskin. These ESI-based ion sources are coupled to an LTQ-Orbitrap analyzer (mass resolving power up to 100,000 m/d(m)) to perform high-resolution mass spectrometry (HRMS) analysis of aerosol filter extracts and of analytes on surfaces (nano-DESI). We perform data processing and mass assignments using custom tools written in Matlab based on a heuristic filtering protocol described by the lab of Oliver Fiehn combined with Kendrick mass defect analysis.

Combustion and thermal-degradation chemistry
 Oxidation also occurs rapidly during the process of combustion or thermal degradation in the presence of air. The burning of biomass (wood, leaves, etc.) is an increasingly common process in the indoor and outdoor atmosphere, and can generate aerosols that are detrimental to human health. Similarly, the smoldering combustion of cigarettes or thermal degradation of glycerol/polyethylene glycol from e-cigarettes may produce aerosols and gases that have adverse health effects. We study biomass burning aerosols that are generated from a custom-built smoldering combustion chamber and smoke particles from cigarette/e-cigarette smoking machines.
Oxidation also occurs rapidly during the process of combustion or thermal degradation in the presence of air. The burning of biomass (wood, leaves, etc.) is an increasingly common process in the indoor and outdoor atmosphere, and can generate aerosols that are detrimental to human health. Similarly, the smoldering combustion of cigarettes or thermal degradation of glycerol/polyethylene glycol from e-cigarettes may produce aerosols and gases that have adverse health effects. We study biomass burning aerosols that are generated from a custom-built smoldering combustion chamber and smoke particles from cigarette/e-cigarette smoking machines.
We investigate the detailed composition of combustion aerosols using HRMS, GC-MS, HPLC-UV, and HPLC-MS. The composition analyses in our lab complement the results from the in-vivo animal inhalation studies and in-vitro cell culture assays from our collaborators (Pinkerton, Denison, Rice).
Other works
Our lab is leading the initiative to develop the first atmospheric chamber database in the United States called ICARUS. This is an NSF-funded collaborative effort between UC Davis and several other institutions (Caltech, NCAR, Carnegie Mellon, UC Irvine, UC Riverside, Georgia Tech, University of Colorado, University of Texas).